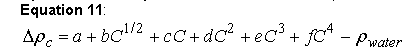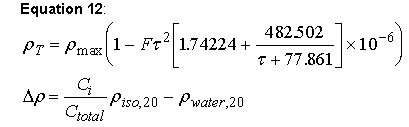Calculating Density
The first step Sednterp uses to calculate the density of a buffer is to calculate the density of the buffer at 20 degrees C. Then, the density is corrected for temperature assuming that water is the predominant component in the buffer. While this is true for solutions containing moderate amounts of other components, significant errors may be introduced for solutions containing high solute concentrations(Ref. 8). Tables of densities of various single solute solutions at various concentrations were fit by least squares methods to a common polynomial form. The sets of coefficients for each solute are stored in the phyconst database. From this, the density of a single solute solution anywhere within the original fit can be interpolated. The density increment of any concentration of buffer component is simply:
where Δρc is the density increment at a certain concentration, a-f are the coefficients from the polynomial fit using least squares methods (Ref. 46), C is the concentration of solute, and ρwater is the density of pure water.
The inclusion of the square root term improves the fit at low concentrations for some ionic solutes. In principle, a is the density of pure water at 20 oC (Ref. 44), but substantially improved fits are obtained when this term is allowed to vary. Thus, equation 11 is used as an empirical fitting function, and, as such, no physical interpretation of the parameters should be made. For each component, the parameters from the most parsimonious polynomial providing an adequate fit are used by Sednterp. Adequacy of the fits were judged by the following criteria: 1) the r.m.s. error of the residuals should correspond to the last decimal place of the tabulated values; and 2) graphs of the residuals as a function of either the dependent or independent variable should exhibit no systematic deviations with magnitudes greater than the fourth decimal place. For alcohols, deviations in the third decimal place were allowed since these data typically are of lower precision.
The concentration range over which each set of coefficients is valid and the maximum error recorded in the phyconst database. In general, polynomials fit the density data for simple salts far better than for organic salts and alcohols. Systematic deviations are observed in fitting ρ for alcohols, with that for ethanol being the worst case. Even though the magnitude of the errors is within the last decimal place of the tabulated data, it is clear that the polynomial function provides only a marginally adequate model for these substances. For most components, the precision of the fit using Sednterp is better than 6*10^(-5) g/ml. An error of this magnitude in ρ will propagate as a 0.3% error in M for a protein having a vbar of 0.75 ml/g and a true M of 130,000. This level of error would be acceptable for most research, but could interfere in more demanding cases (e.g. determining association constants for cases where large stoichiometries are observed).
Sednterp also calculates a density increment for the presence of isotopes of oxygen and hydrogen in the water of the buffer. This increment is calculated using the methods described by Steckel and Szapiro (Ref. 45). Equation 12 is used both for the density increment at 20 degrees C and to compute the temperature correction factor.
where ρT is the density at temperature T in C, ρmax is the maximum density (observed at the maximum temperature), τ is the temperature difference between that of the experiment and the temperature of maximum density, F is an empirical constant, is the concentration of one isotope and C-total is the concentration of all the isotopes and the densities of the isotopes and plain water are computed at 20 C. Equation 12 may be used for all water species by substituting in the appropriate values from the following table. Note that in cases where O-18 is present, isotopic purity is about 98%, and it is 100% when D is present. Values for H2O reflect the natural isotopic abundance of O-18 and D. Likewise, O and H reflect natural isotopic abundance in D2O and H2O18. Equation 12 is valid from the melting temperature to approximately 80 C. Simple mixing rules are used when different isotopes of water are combined. (Ref. 45) The values used in equation 12 are loaded in the phyconst database and are listed here also: